|
12/29/2023 0 Comments Change safari shortcuts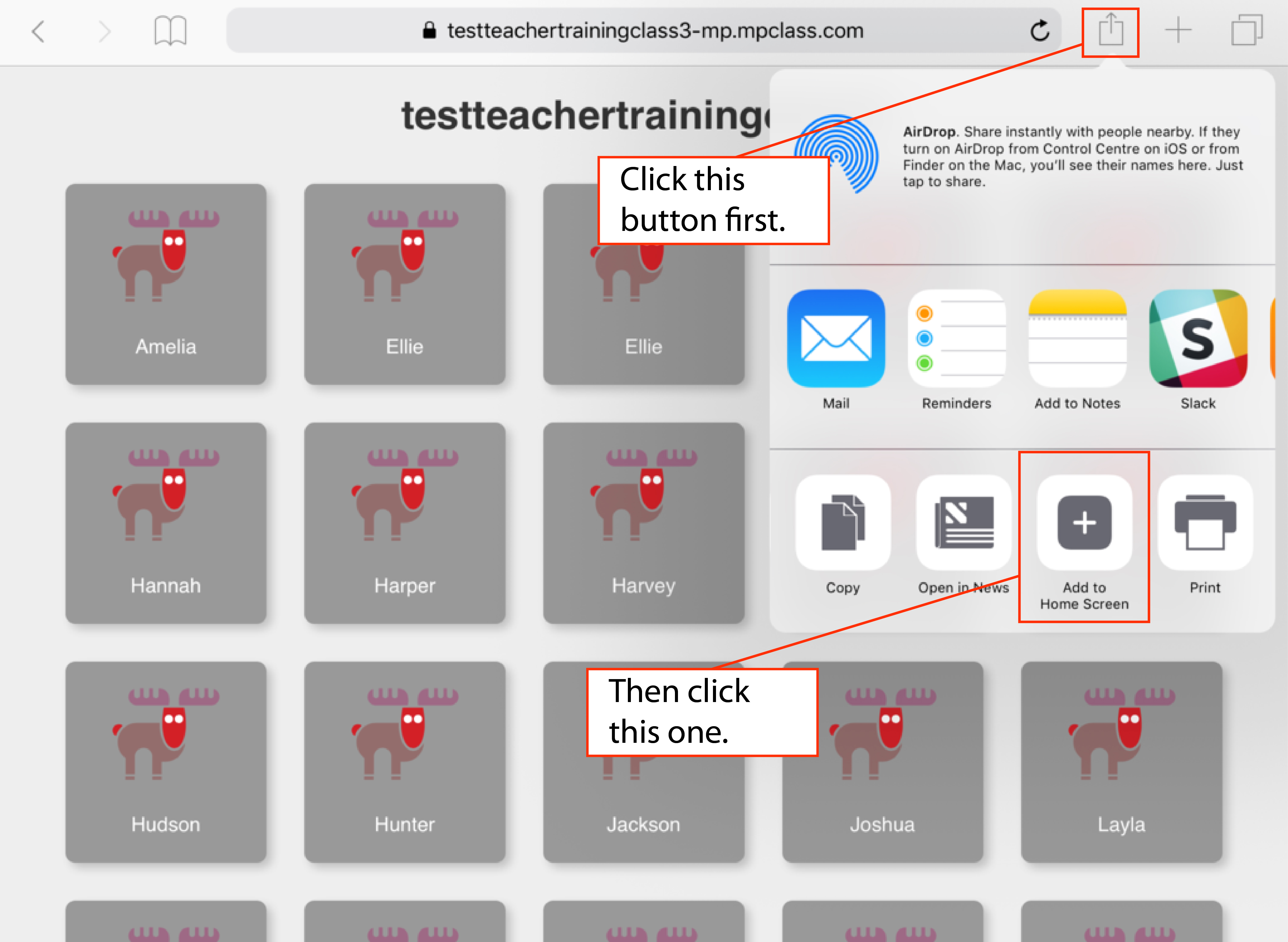
Have the ability to work off-shift and extra hours as requiredĭoes this sound like you? If so, please apply today!.Comfortable in a fast-paced small company environment with minimal direction and able to adjust workload based upon changing priorities.Excellent interpersonal, verbal and written communication skills that are critical in this collaborative work environment.Ambitious and willing to accept temporary responsibilities outside of initial job description.Ability to think critically with validated troubleshooting and analytical skills.You have skills in Microsoft Word, Excel and data analysis.You have experience in handling and propagation of human primary cells, including T cells.You have experience with automated processing equipment.You possess excellent aseptic/sterile techniques and prior experience with cell culture, cell culture equipment including rocker bag bioreactors.You rigorously adhere to SOPs and cGMP regulations, with the ability to accurately complete documentation associated with commercial manufacturing.You have confirmed knowledge of pharmaceutical manufacturing of biotechnology products, aseptic processing, cell culture and/or vector products.Previous experience with cell therapy or vector products preferred.You have cell culture processing experience.You have a Bachelor’s degree and/or advanced degree with 3 or more years of cGMP experience in a manufacturing pharmaceutical/biotechnology environment.Requirements - we would love to hear from you if: Successfully troubleshoot processing and equipment issues.Assist in the development of SOPs, batch records, deviations and change controls.Work as part of a team to execute GMP runs in close collaboration with Process Development and Quality.Perform all tasks associated with the manufacture of clinical product following batch records and standard operating procedures (SOPs) to ensure safe and compliant operations.Responsibilities include, but are not limited to: Under minimal supervision, you will be accountable for and focused on front line manufacturing of our clinical stage cell therapies. The facility is being developed as a Center of Excellence for the advancement of novel TCR mediated T cell therapies targeting tumor mutations and antigens. We are seeking a self-motivated and ambitious individual with strong work ethic and leadership qualities to join us as a Cell Therapy Specialist at our clinical production facility in Gaithersburg, Maryland. Every single day, we seek to establish a direct line between that purpose and our day-to-day work. Please log onto your Internal Career Site to apply for this jobĮveryone at Kite is grounded by one common goal – curing cancer. For Current Kite Pharma Employees and Contractors:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
